Fezolinetant explained
- Fezolinetant is a new drug that has been approved to treat hot flushes and night sweats
- This article summarises safety and efficacy studies of fezolinetant
- It also looks at how fezolinetant works, side effects and interactions with other medications and supplements
What is fezolinetant?
Fezolinetant – brand name Veoza – is a new drug that has been recently approved in the UK to treat moderate to severe vasomotor symptoms in menopausal women aged 45 to 60 years [1]. It has also been approved in the United States, Europe, and several other countries.
Up to 80% of perimenopausal and menopausal women experience hot flushes and night sweats (also known as vasomotor symptoms), which can disrupt sleep and adversely affect mood. Other common menopausal symptoms include fatigue, memory problems, low mood, anxiety, poor sleep, headaches, muscle and joint pains, and urinary symptoms. It is estimated that at least 1 in 10 menopausal women experience severe symptoms that have a negative impact on their relationships and ability to work and function.
How does fezolinetant work?
Vasomotor symptoms are thought to be caused by increased activity in kisspeptin, neurokinin B (NKB), and dynorphin neurones, or KNDy neurones (pronounced ‘candy’ neurones), in the thermoregulatory centre of the brain. Kisspeptin, neurokinin B, and dynorphin, are all neurotransmitters which are chemicals made by the body that allow nerves to communicate with each other. Normally there is a balance between neurokinin B, which upregulates activity in KNDy neurones; and oestrogen, which downregulates activity. During menopause oestrogen levels fall leading to increased activity in the KNDy neurones. The thermoregulatory centre becomes hypersensitive, and the body overreacts to a mild increase in external temperature by sweating and flushing in an attempt to cool down.
Fezolinetant works by blocking neurokinin 3 receptors (NK3R) that NKB normally binds to. This prevents the KNDy neurones becoming overstimulated and calms down activity in the thermoregulatory centre.
KNDy neurones are also central to the hormonal control of the menstrual cycle and reproduction. They control the release of gonadotrophin releasing hormone (GnRH) from the hypothalamus, an area in the brain that links the endocrine system with the nervous system. NKB stimulates the release of GnRH which drives ovarian function and the production of oestrogen, progesterone, and testosterone from the ovaries [2]. Neurokinin B receptors are also found in other locations in the central nervous system, as well as the ovaries and the womb [3]. The effects of blocking NK-B receptors elsewhere in the brain and body have not been fully assessed.
Fezolinetant is currently only available on private prescription in the UK, but a NICE Technology Appraisal is underway and it is likely that fezolinetant will be available in the NHS later this year, if approved.
How effective is fezolinetant?
The efficacy of fezolinetant has been tested in 1022 postmenopausal women in two 12-week trials [4,5]. Compared with placebo, women treated with fezolinetant experienced 2.5 fewer hot flushes and night sweats over a 24-hour period, and the severity was significantly reduced. The effects were modest, but women with more frequent and/or severe symptoms are likely to reap greater benefit.
What are the side effects associated with fezolinetant?
The safety of fezolinetant has been assessed in 2203 women in three clinical trials of up to 12 months duration (4, 5, 6). The most common side effects reported by women taking fezolinetant were abdominal pain, diarrhoea, and insomnia [1]. Some 2.1% of women taking fezolinetant developed signs of liver damage (elevated levels of liver enzymes) that resolved when the drug was stopped.
In October 2023, a paper was published in the Lancet that reported an increased incidence of cancer in women treated with fezolinetant compared with placebo [7]. When data was combined from the three clinical trials, there were two cases of cancer in 952 women assigned to placebo (which is in line with the annual cancer incidence in women of a similar age in the general population), seven cases of cancer in 1103 women assigned to fezolinetant 30mg daily, and sixteen cases of cancer in 1100 women assigned to fezolinetant 45mg daily. Overall, women treated with fezolinetant were 2.8 to 4.3 times more likely to receive a cancer diagnosis across the 12-month period, depending on the dose received.
The following graph illustrates the number of women in each group who were diagnosed with cancer during the 12-month study period [8]. The recommended dose to treat vasomotor symptoms is 45mg once daily. Women took fezolinetant for three months and were monitored for up to 12 months.
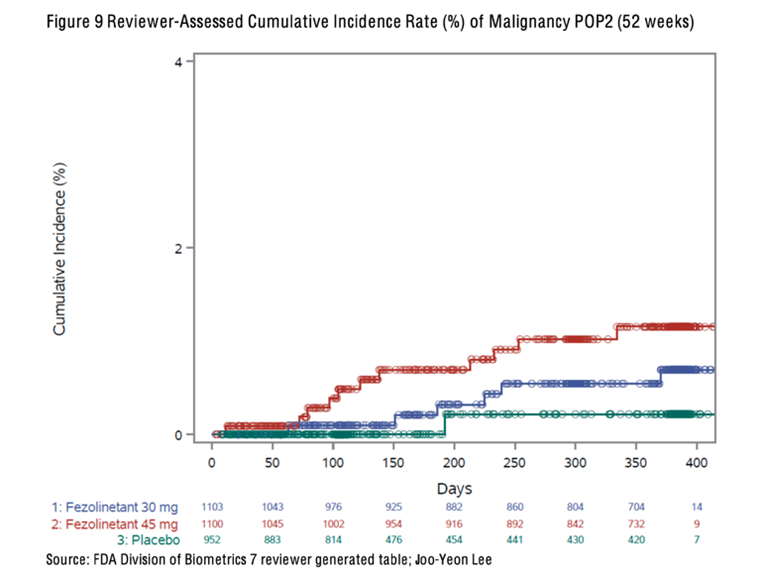
This data does not prove that fezolinetant causes cancer. Two cases occurred within a month of the start of the trials, and it is not feasible that a drug could cause cancer in such a short time. Even 12 months is a very short time period for cancer to develop. However, the higher incidence of cancer in women taking fezolinetant in clinical trials requires further evaluation, and more information is needed to determine whether a causal relationship exists.
What does all this mean?
The most effective evidence-based treatment for perimenopausal and menopausal symptoms, including vasomotor symptoms, is hormone replacement therapy (HRT), because HRT targets the underlying cause. Further, when initiated within 10 years of the menopause HRT is bone, heart and neuro-protective, and reduces all-cause mortality by 30% [9]. You can download visual aids illustrating the benefits and risks of HRT here.
Fezolinetant and cancer
Fezolinetant is a non-hormonal treatment option for vasomotor symptoms and may be especially useful for women who can’t have HRT, such as some women with a history of oestrogen receptor positive breast cancer. Women receiving treatment for cancer, including breast cancer, have not been included in any of the clinical studies, and fezolinetant is not currently recommended for use in these women as the safety and efficacy are unknown.
A decision to treat women with a past history of cancer should be based on an individualised assessment of the relative risks and benefits [1]. To enable women to make an informed treatment choice they should be aware that the available safety data is limited, and an increased incidence of cancer was observed in clinical trials that has not yet been fully evaluated. Until further information is available, women may prefer to explore other treatment options for hot flushes and night sweats, such as cognitive behavioural therapy, or antidepressants.
Fezolinetant and liver function
Routine monitoring of liver function in women taking fezolinetant is not currently recommended in the UK. However, 2.1% of women who took fezolinetant in clinical trials developed abnormal liver function, which is substantially higher than the number of women who develop abnormal liver function when taking statins to lower cholesterol [10], or an antifungal medication called terbinafine [11], and monitoring of liver function is advised for both. It’s not clear why the advice regarding fezolinetant is different, particularly as the data is limited and most women who developed abnormal liver function in the trials had no symptoms. If these women had continued to take fezolinetant it is possible that the inflammation in the liver would have progressed and eventually made them unwell. Clinicians are advised to discontinue fezolinetant in women with abnormal liver function and wait for liver enzymes to normalise, but they won’t be aware of a change if women are not being monitored.
Fezolinetant and testosterone
In men, fezolinetant significantly reduces blood testosterone levels [12]. When used at higher doses (60-180mg daily), fezolinetant has also been shown to inhibit ovulation and suppress testosterone in premenopausal women with polycystic ovarian syndrome (PCOS) [13]. Fezolinetant has not been shown to reduce testosterone levels in postmenopausal women with vasomotor symptoms, but the trials did not include perimenopausal women with regular or irregular periods who may also experience VMS. It is important that perimenopausal women with VMS are aware that fezolinetant may suppress testosterone, because testosterone deficiency causes or contributes to many menopausal symptoms such as low mood, VMS, headaches, low libido, brain fog, joint pain, and vaginal dryness [14].
Fezolinetant interactions with other medications and supplements
Finally, fezolinetant is metabolised in the liver by an enzyme known as CYP1A2. Drugs that are moderate or strong inhibitors of CYP1A2 may increase blood levels of fezolinetant and should be avoided. Examples include ciprofloxacin, an antibiotic; fluvoxamine, an antidepressant; and ethinyl oestradiol, an oral oestrogen in many types of contraceptive pill and some oral HRT formulations. Liquorice, and certain supplements such as St John’s Wort, which is often taken to treat low mood symptoms, and green tea and grape seed extract, which are often taken for their anti-inflammatory properties, should also be avoided.
Taking fezolinetant at the same time as HRT has not been studied, and it is therefore not recommended to use fezolinetant and HRT together [1].
Conclusion
Fezolinetant is a new drug that has been approved to treat moderate to severe vasomotor symptoms in menopausal women. Fezolinetant does not treat other menopausal symptoms, and we don’t yet have long-term data regarding the impact of fezolinetant on cardiovascular and bone health, or breast cancer incidence.
Experts have concluded that: ‘a new option to treat bothersome menopausal symptoms definitely represents a step forward. Although there will be a clinical role for NK3R antagonists, gaps remain in ascertaining the precise balance of benefit/risk, especially in women with contraindications or at high risk for HRT who could benefit most from this new addition’ (15).
It is imperative that women are counselled about the benefits and the risks – both the known and potential risks – and given comprehensive information about all the available treatment options, to enable them to make an informed treatment decision.
Written by Dr Sarah Glynne and Dr Louise Newson
References
1. Electronic medicines compendium (2023) Veoza 45mg film-coated tablets. Astellas Pharma Ltd, https://www.medicines.org.uk/emc/product/15361/smpc#gref
2. Lehman M.N., Coolen L.M., Goodman R.L. (2010), ‘Minireview: kisspeptin/neurokinin B/dynorphin (KNDy) cells of the arcuate nucleus: a central node in the control of gonadotropin-releasing hormone secretion’, Endocrinology, 151(8):3479-89. doi:10.1210/en.2010-0022
3. Patak E., Candenas M.L., Pennefather J.N., Ziccone S., Lilley A., Martin J.D., et al. (2003), ‘Tachykinins and tachykinin receptors in human uterus’, Br J Pharmacol, 139(3):523-32. doi:10.1038/sj.bjp.0705279
4. Lederman S., Ottery F.D., Cano A., Santoro N., Shapiro M., Stute P., et al. (2023) ‘Fezolinetant for treatment of moderate-to-severe vasomotor symptoms associated with menopause (SKYLIGHT 1): a phase 3 randomised controlled study’, Lancet, 401(10382):1091-102. doi.org/10.1016/S0140-6736(23)00085-5
5. Johnson K.A., Martin N., Nappi R.E., Neal-Perry G., Shapiro M., Stute P., et al. (2023), ‘Efficacy and safety of fezolinetant in moderate to severe vasomotor symptoms associated with menopause: a Phase 3 RCT’, J Clin Endocrinol Metab, 108(8):1981-97. doi:10.1210/clinem/dgad058
6. Neal-Perry G., Cano A., Lederman S., Nappi R.E., Santoro N., Wolfman W., et al. (2023), ‘Safety of fezolinetant for vasomotor symptoms associated with menopause: a randomized controlled trial’, Obstet Gynecol, 141(4):737-47. doi:10.1097/AOG.0000000000005114
7. Douxfils J., Beaudart C., Dogne J.M. (2023), ‘Risk of neoplasm with the neurokinin 3 receptor antagonist fezolinetant’, Lancet, 402(10413):1623-5. doi.org/10.1016/S0140-6736(23)01634-3
8. FDA Centre for Drug Evaluation and Research. Application number: 216578Orig1s000 CLINICAL REVIEW(S). January 23, 2023. Available at: https://www.accessdata.fda.gov/drugsatfda_docs/nda/2023/216578Orig1s000MedR.pdf
9. Langer R.D. (2021), ‘The role of medications in successful aging’, Climacteric, 24(5):505-12. doi:10.1080/13697137.2021.1911991
10. Hippisley-Cox J., Coupland C. (2010), ‘Unintended effects of statins in men and women in England and Wales: population based cohort study using the QResearch database’, BMJ, 340:c2197.
10. Hippisley-Cox J., Coupland C. (2010), ‘Unintended effects of statins in men and women in England and Wales: population based cohort study using the QResearch database’, BMJ, 340:c2197. doi.org/10.1136/bmj.c2197
11. NIH. U.S. National Library of Medicine. LiverTox: clinical and research information on drug-induced liver injury. Bethesda (MD): National Institute of Diabetes and Digestive and Kidney Diseases; 2012-. Terbinafine. [Updated 2018 Jan 1]. Available at: https://www.ncbi.nlm.nih.gov/books/NBK548617/pdf/Bookshelf_NBK548617.pdf
12. Fraser G.L., Ramael S., Hoveyda H.R., Gheyle L, Combalbert J. (2016), ‘The NK3 receptor antagonist ESN364 suppresses sex hormones in men and women’, J Clin Endocrinol Metab, 101(2):417-26. doi:10.1210/jc.2015-3621
13. Fraser G.L, Obermayer-Pietsch B., Laven J., Griesinger G., Pintiaux A., Timmerman D., et al. (2021), ‘Randomized controlled trial of neurokinin 3 receptor antagonist fezolinetant for treatment of polycystic ovary syndrome’, J Clin Endocrinol Metab, 106(9):e3519-e32. doi: 10.1210/clinem/dgab320
14. Davis S.R. (2013), ‘Androgen therapy in women, beyond libido’, Climacteric,16 Suppl 1:18-24. doi:10.3109/13697137.2013.801736
15. Gompel A., Stuenkel C.A. (2023), ‘Neurokinin 3 receptor antagonists for menopausal vasomotor symptoms, an appraisal’, Cell Rep Med, 4(6):101076. doi.org/10.1016/j.xcrm.2023.101076





